Reducing Blood Culture Contamination in the ED: Impact of Kurin Lock® Implementation
Browne E, Russell M, Muniyappa GK, Abbas M, Ratenavelu P, Fitzpatrick F, Keane O, McDermott H.
Beaumont Hospital, Dublin, Ireland.
Infection Prevention in Practice.
Background
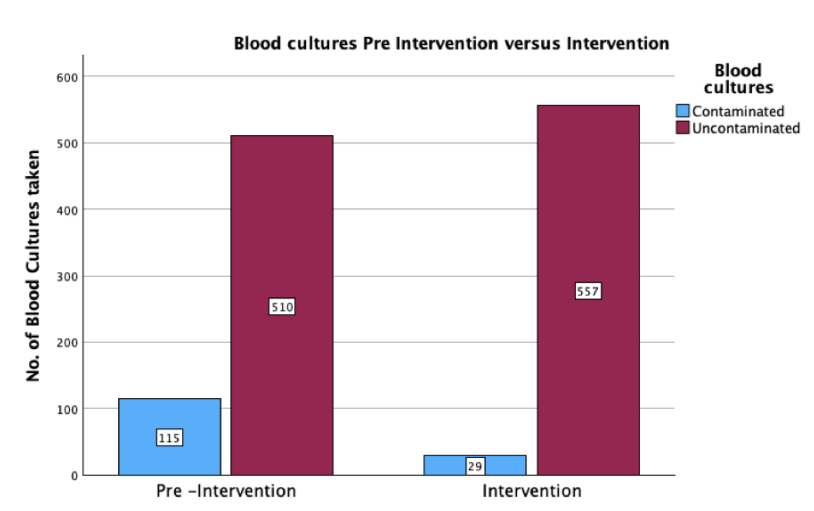
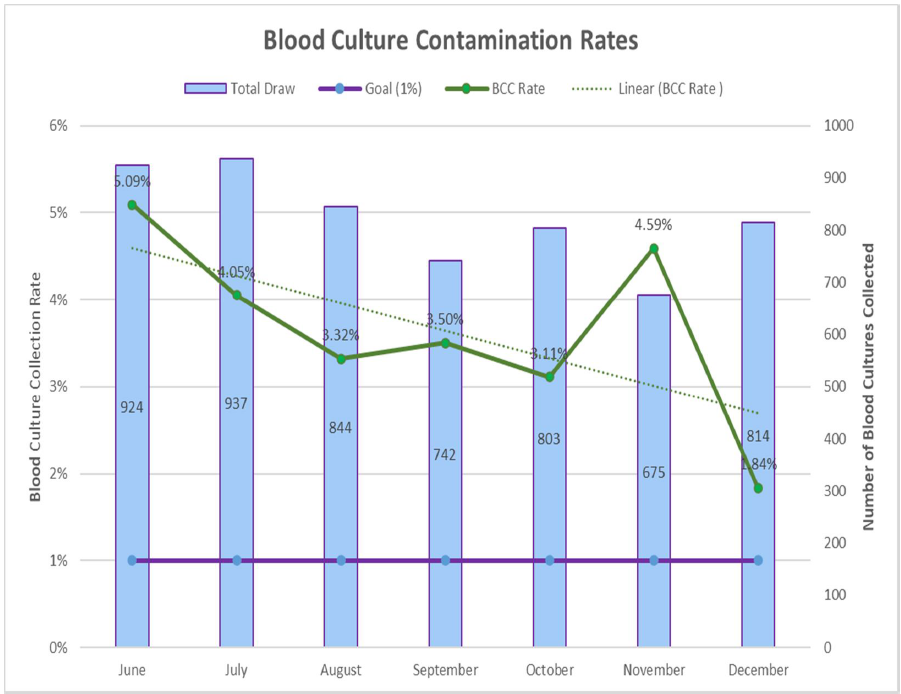
Blood culture contamination (BCC) poses significant clinical and financial challenges in hospital settings. Guidelines recommend BCC rates remain below 3%; however, in Q1 2024, our tertiary hospital emergency department (ED) reported a median rate of 10%.
Aim
To evaluate the impact of the Kurin Lock® device on reducing BCC rates in the ED. The device diverts the initial 0.15 ml of blood, potentially containing skin flora contaminants.
Methods
An eight-week pilot of the Kurin Lock® device was conducted from May to July 2024. ED clinical staff received training prior to implementation. Blood cultures collected using Kurin Lock® were compared with those obtained via the standard method. Contamination was defined by the Clinical Microbiology Team as isolation of organisms considered clinically insignificant and likely derived from skin flora. Data were extracted from the hospital’s surveillance system.
Findings
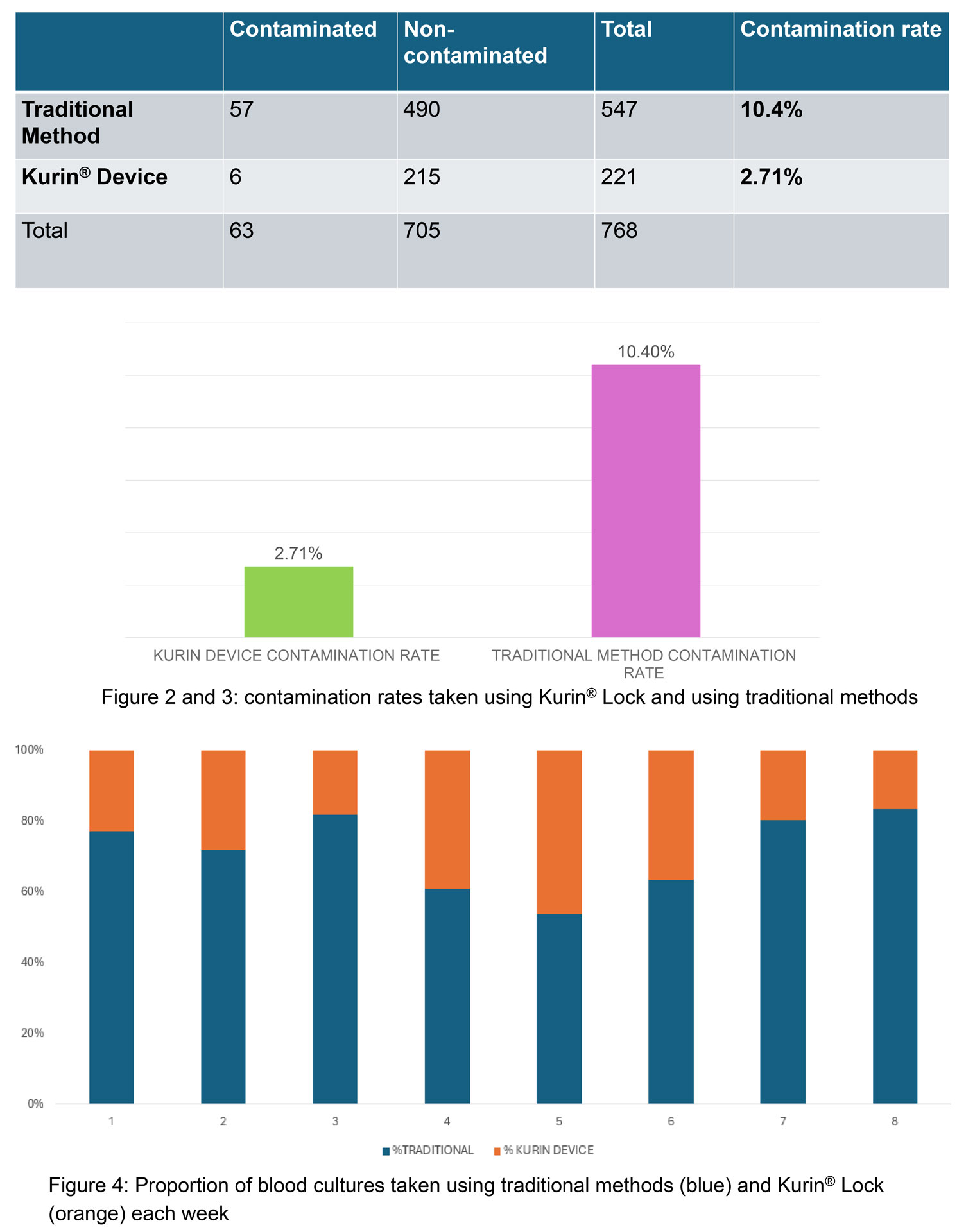
A total of 768 blood cultures were collected. Contamination occurred in 2.7% (n=6/221) blood cultures obtained using Kurin Lock® compared with 10.4% (n=57/547) blood cultures obtained using the standard method (p=0.0004), representing a 74% relative reduction. Staff adherence to device use averaged 28.5%.
Conclusion
Use of the Kurin Lock® was associated with a significant reduction in BCC, achieving rates below the 3% recommended standard. These findings highlight the potential benefits for improving patient outcomes and healthcare costs. Ongoing success will depend on increasing staff engagement and integration into routine practice.
Read full text here