We and selected third parties use cookies or similar technologies as specified in our privacy policy. You can consent to the use of such technologies by closing this notice, by interacting with any link or button outside of this notice or by continuing to browse otherwise.
AcceptLearn moreWe may request cookies to be set on your device. We use cookies to let us know when you visit our websites, how you interact with us, to enrich your user experience, and to customize your relationship with our website.
Click on the different category headings to find out more. You can also change some of your preferences. Note that blocking some types of cookies may impact your experience on our websites and the services we are able to offer.
These cookies are strictly necessary to provide you with services available through our website and to use some of its features.
Because these cookies are strictly necessary to deliver the website, refusing them will have impact how our site functions. You always can block or delete cookies by changing your browser settings and force blocking all cookies on this website. But this will always prompt you to accept/refuse cookies when revisiting our site.
We fully respect if you want to refuse cookies but to avoid asking you again and again kindly allow us to store a cookie for that. You are free to opt out any time or opt in for other cookies to get a better experience. If you refuse cookies we will remove all set cookies in our domain.
We provide you with a list of stored cookies on your computer in our domain so you can check what we stored. Due to security reasons we are not able to show or modify cookies from other domains. You can check these in your browser security settings.
These cookies collect information that is used either in aggregate form to help us understand how our website is being used or how effective our marketing campaigns are, or to help us customize our website and application for you in order to enhance your experience.
If you do not want that we track your visit to our site you can disable tracking in your browser here:
We also use different external services like Google Webfonts, Google Maps, and external Video providers. Since these providers may collect personal data like your IP address we allow you to block them here. Please be aware that this might heavily reduce the functionality and appearance of our site. Changes will take effect once you reload the page.
Google Webfont Settings:
Google Map Settings:
Google reCaptcha Settings:
Vimeo and Youtube video embeds:
The following cookies are also needed - You can choose if you want to allow them:
You can read about our cookies and privacy settings in detail on our Privacy Policy Page.
Privacy Policy
Reducing Blood Culture Contamination in the ED: Impact of Kurin Lock® Implementation
/in Clinical StudiesBrowne E, Russell M, Muniyappa GK, Abbas M, Ratenavelu P, Fitzpatrick F, Keane O, McDermott H.
Beaumont Hospital, Dublin, Ireland.
Infection Prevention in Practice.
Background
Blood culture contamination (BCC) poses significant clinical and financial challenges in hospital settings. Guidelines recommend BCC rates remain below 3%; however, in Q1 2024, our tertiary hospital emergency department (ED) reported a median rate of 10%.
Aim
To evaluate the impact of the Kurin Lock® device on reducing BCC rates in the ED. The device diverts the initial 0.15 ml of blood, potentially containing skin flora contaminants.
Methods
An eight-week pilot of the Kurin Lock® device was conducted from May to July 2024. ED clinical staff received training prior to implementation. Blood cultures collected using Kurin Lock® were compared with those obtained via the standard method. Contamination was defined by the Clinical Microbiology Team as isolation of organisms considered clinically insignificant and likely derived from skin flora. Data were extracted from the hospital’s surveillance system.
Findings
A total of 768 blood cultures were collected. Contamination occurred in 2.7% (n=6/221) blood cultures obtained using Kurin Lock® compared with 10.4% (n=57/547) blood cultures obtained using the standard method (p=0.0004), representing a 74% relative reduction. Staff adherence to device use averaged 28.5%.
Conclusion
Use of the Kurin Lock® was associated with a significant reduction in BCC, achieving rates below the 3% recommended standard. These findings highlight the potential benefits for improving patient outcomes and healthcare costs. Ongoing success will depend on increasing staff engagement and integration into routine practice.
Read full text here
Category Leader Kurin, Inc. Reports Record Revenue for Q1 2026
/in Featured, Press ReleasesKurin, Inc., the inventor and manufacturer of Kurin® blood culture collection sets, announced continued strong revenue growth and record revenues for the first quarter of 2026. Building on its earlier trajectory, the company has achieved significant year-over-year expansion, driven by increasing adoption across leading healthcare systems despite ongoing industry-wide financial pressures.
“Our performance reflects acceleration and not simply resilience,” said Bob Rogers, CEO of Kurin, Inc. “Health systems are under more pressure than ever to improve outcomes while reducing costs. Kurin delivers both, providing measurable clinical and economic impact without adding complexity to frontline workflows.”
Since 2017, Kurin has expanded its presence across hospitals nationwide as blood culture contamination has become a defined quality and cost metric. Increasing alignment with regulatory and advisory bodies is accelerating adoption, driving consistent revenue growth, and deepening health system engagement for solutions that reduce the preventable costs of blood culture contamination.
“To support this next phase, we have invested in our commercial infrastructure, operations, and manufacturing capacity,” Rogers added. “We are expanding and leading this rapidly growing category as standards of care are quickly evolving.”
Kurin’s proprietary technology simplifies blood culture collection best practices by automatically diverting potential contaminants without adding steps for clinicians. Kurin’s evidence-based, proven technology is uniquely valuable as health systems intensify efforts to reduce avoidable costs, prevent diagnostic errors, and limit unnecessary escalation of care.
About Kurin:
Kurin® is a privately held, certified minority-owned business dedicated to engineering better healthcare through innovative, cost-effective, clinician-approved technologies. Building on the successful invention, clinical adoption, and corporate acquisition of CUROS™ disinfecting port protectors, Kurin is focusing its depth of experience in medical device engineering, marketing, sales, and service to tackle the persistent and costly fallout of false positive blood cultures. With agile development capabilities and intense customer focus, the Kurin Blood Collection Systems have quickly gained caregiver acceptance, have been proven to sideline skin contaminants during blood culture collection, and have become the market-leading solution for blood culture collection best practice.
CUROS™ is a registered trademark of Solventum and Kurin has no connection to Solventum.
Kurin Prevails in Federal Appellate Court in Long-Running Litigation with Magnolia Medical Technologies
/in Featured, Press ReleasesKurin, Inc., a leader in innovative blood culture collection technology designed to reduce contamination and improve patient outcomes, announced today that the United States Court of Appeals for the Federal Circuit has ruled in Kurin’s favor in a patent dispute brought by Magnolia Medical Technologies accusing Kurin’s Lock of infringement. The U. S. District Court had previously ruled in Kurin’s favor and the appellate court affirmed the district court’s judgment of non-infringement.
“Although we expected this ruling, it is gratifying to see another Court validate our position that Kurin Lock does not infringe Magnolia patents, as they had claimed. This ruling affirms the strength and independence of Kurin’s technology,” said Bob Rogers, Chairman of the Board and CEO of Kurin, Inc. “We have always been confident in the innovation behind Kurin Blood Culture Collection Sets and their ability to help clinicians reduce blood culture contamination. Our focus remains on advancing solutions that improve patient care while helping hospitals—already facing significant resource constraints—reduce unnecessary costs.”
Blood culture contamination is a well-recognized challenge in healthcare that can lead to false-positive results, unnecessary treatments, and increased healthcare costs. Kurin’s solution addresses this problem by sidelining the initial portion of a blood draw that is most likely to contain skin contaminants, helping clinicians obtain more accurate diagnostic samples.
Kurin will continue to focus on innovation, clinical impact, and supporting healthcare providers with solutions that improve diagnostic accuracy and patient outcomes.
About Kurin:
Kurin® is a privately held, certified minority-owned business dedicated to engineering better healthcare through innovative, cost-effective, clinician-approved technologies. Building on the successful invention, clinical adoption, and corporate acquisition of CUROS™ disinfecting port protectors, Kurin is focusing its depth of experience in medical device engineering, marketing, sales, and service to tackle the persistent and costly fallout of false positive blood cultures. With agile development capabilities and intense customer focus, the Kurin Blood Collection Systems have quickly gained caregiver acceptance, have been proven to sideline skin contaminants during blood culture collection, and have become the market-leading solution for blood culture collection best practice.
CUROS™ is a registered trademark of Solventum and Kurin has no connection to Solventum.
Beyond the needle: innovative approaches for reducing blood culture contamination rates in the emergency department
/in Clinical StudiesKenny S, Raouf Z, Sheridan A, McCarthy I, Rothwell-Kelly G, Menzies D, Schaffer K.
St. Vincent’s University Hospital, Dublin, Ireland
Ir Med J. 2025:118(8); P142
Aim
This quality improvement project aimed to evaluate the effectiveness of the Kurin Lock device, which diverts the initial 0.15 mL of blood, and ChloraPrep (2% alcoholic chlorhexidine) in reducing blood culture contamination rates in emergency departments.
Methods
The study was conducted across three phases: pre-intervention, intervention, and postintervention. Before the intervention, alcohol or chlorhexidine wipes were used for skin antisepsis. The intervention phase introduced the Kurin Lock device and ChloraPrep, accompanied by staff training. Contamination rates were analyzed using the chi-square test.
Results
During the intervention phase, contamination rates significantly decreased from 18.4% (preintervention) to 4.9% (p < 0.001). However, in the post-intervention phase, contamination rates rebounded to 18.1%.
Discussion
The study demonstrates that combining specimen diversion systems with enhanced antisepsis protocols effectively reduces blood culture contamination. However, maintaining these improvements over time remains a challenge, emphasizing the need for sustained adherence to the intervention.
Excerpt: “Blood specimen diversion systems, such as the Kurin Lock device, effectively reduce contamination by diverting the initial blood sample likely to contain skin contaminants, a method well-supported in the literature. A recent review further highlights their role in improving diagnostic precision.[Al Mohajer M and Lasco T. Open Forum Infect Dis. 2023] In April 2024 the National Institute for Health and Care Excellence (NICE) issued Medical Technologies Guidance 77, which recommends blood diversion systems such as Kurin Lock for EDs with contamination rates > 2.4 %, providing a strong policy mandate for their use.”
Read full text here
Improving Blood Culture Contamination in an Adult Emergency Department Utilizing Sideline Technology
/in Clinical StudiesLesly Evert, BSN, RN, CEN, EMT-P and Tina Nielson, RN, MS, CEN, ACNS-BC, APNP
Froedtert Hospital, Milwaukee, WI
Emergency Nursing 2025
Purpose
The purpose of this project was to decrease the BCC rate in an Academic Medical Center and expand knowledge about adherence to best practice to positively affect patient outcomes.
Problem
Emergency Department blood culture contaminations rate reported at 6%.
Blood culture contamination (BCC) can lead to:
Gap Analysis:
Clinical Question
In an adult Emergency Department, does skin prep and wasting blood before specimen collection using a specimen diversion device (ISDD) reduce contamination rates of blood cultures to < 1%?
Setting and Sample
Design
An evidenced-based practice project utilizing the John Hopkins-Evidence Based Practice Model
Methods
Educational Strategies:
Learning Objectives:
Data collected:
Results
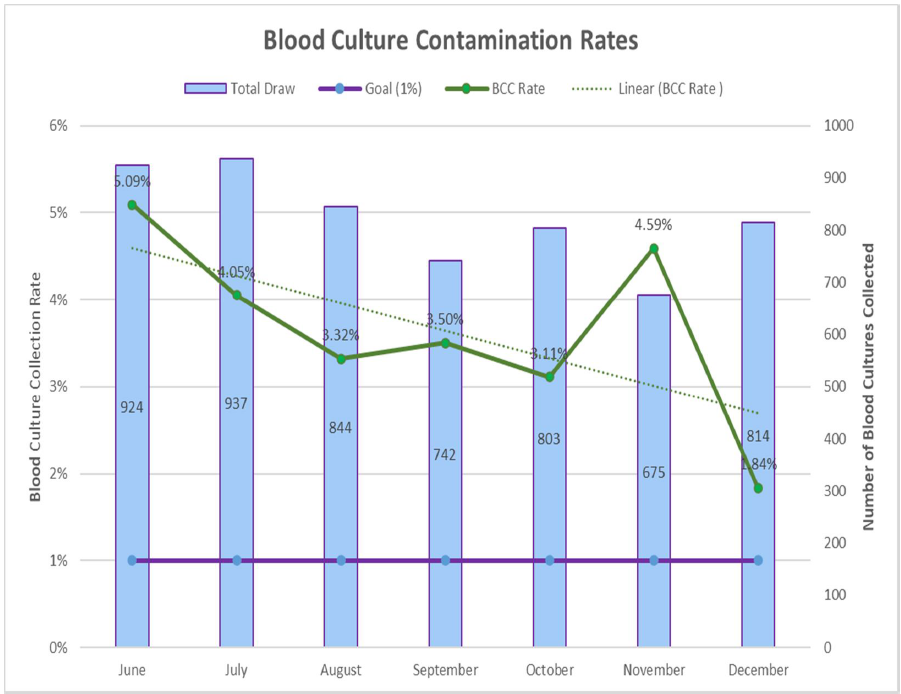

Findings/Outcomes
Results
Conclusions
This project demonstrated that a multimodal approach improved BCC rates. Methods include:
A limitation of the project was we were unable to account for compliance with ISDD utilization.
Implications for Practice
Standardized education and implementation of ISDD for waste diversion should be initiated for consistent blood culture collection.
Acknowledgement
ED Shared Governance, nursing staff, leadership and lab for their support and ongoing focus to providing quality patient care
For additional information please contact:
Lesly Evert, BSN, RN, CEN, EMT-P
Emergency Department, Froedtert Hospital
Lesly.Evert@froedtert.com
Read full text here
Analysis of the Clinical Significance of Positive Blood Cultures in the Emergency Department: A Single-Center Study
/in Clinical StudiesKarlath FA, Rehan MA, Geigor A, Mitchell J, Arnaout S, Greenough TC, and Elliso RT.
UMass Memorial Health
Open Forum Infectious Disease June 17, 2025
Background
There have been major advances in blood culture technology in the last decade with both faster and more sensitive pathogen detection as well as more precise species identification. We have performed a reassessment of the results of positive blood cultures in this new clinical microbiology era with a focus on contaminant identification.
Methods
A retrospective study was conducted including all patients with a blood culture collected in two UMass Memorial Health emergency departments from September 2019 through April 2020. Contaminants were identified by standard clinical microbiology lab criteria and by independent retrospective review by 3 infectious disease (ID) physicians and an Infectious Disease fellow.
Results
5,673 blood samples were obtained with 5,661 samples analyzed after 12 were deemed inconclusive by the ID physician review. Blood culture contaminants accounted for 22.5% of the positive blood cultures. Staphylococcus epidermidis was the most frequent contaminant (33.4%) while Escherichia coli was the most frequent pathogen (21%) causing true bacteremia. Coagulasenegative staphylococci remain the most frequent cause of blood culture contamination with Staphylococcus epidermidis being the most common. Staphylococcus auricularis, Staphylococcus caprae, lentus, Staphylococcus pseudointermedius, Staphylococcus saccharolyticus, and Staphylococcus warneri were all determined to be contaminants in 100% of cases.
Relevant Conclusions
The results of the prospective controlled trial associated with this study affirm that the blood diversion device did lead to a decrease in blood culture contamination rates in busy ED in both the intention-to-use and actual-use analyses. “These devices offer a cost-effective and clinically beneficial strategy to improve diagnostic accuracy and patient outcomes. We anticipate that this approach could help combat the issue of increasing antibiotic resistance.”
Read full text here
Reducing Blood Culture Contamination Rates Below 1%: A Strategic Approach
/in Clinical StudiesSamantha Biddle, Jo Whitis
IU Health, Indiana
Clinical Nurse Specialist 39(4):p 192-194, 7/8 2025
Subscription required
Impact of Initial Specimen Diversion Technique on Blood Culture Contamination Rates
/in Clinical StudiesDhammika Navarathna, Janell Lukey
Veterans Affairs Central Texas Healthcare System
Fed Pract. 2025;42(6):e0596. June 17. doi:10.12788/fp.0596
Background
A false-positive blood culture contamination rate of 3.0% is a widely recognized hospital benchmark. In November 2017, the Veterans Affairs Central Texas Healthcare System (VACTHCS), a 146-bed urban teaching hospital with about 30,000 annual emergency department (ED) visits, introduced the initial specimen diversion technique (ISDT) to reduce its contamination rate. ISDT uses blood draws to isolate and discard the initial portion of blood collected, known to be more prone to contamination from skin bacteria.
Methods
This retrospective comparative study of ISDT effectiveness was conducted at VACTHCS. Data were collected 36 months before and after ISDT implementation. Adults admitted to VACTHCS through the ED who required blood cultures for clinical suspicion of infection were included. Data were reviewed 36 months postimplementation to determine the effectiveness of the ISDT intervention.
Results
There was a marked decrease in contamination rates within the VACTHCS ED. Pre-implementation, the mean contamination rate was 4.5% (95% CI, 3.90-4.90), which decreased to 2.6% (95% CI, 2.10-3.20) following ISDT implementation (P < .001).
Conclusions
Targeted interventions such as ISDT may reduce blood culture contamination rates in the ED. Incorporating ISDT at VACTHCS resulted in an overall blood culture contamination reduction and improved patient care.
Read full text here
Kurin and Crouse Hospital: A Transformative Partnership in Reducing Contaminated Blood Cultures
/in Featured, Press ReleasesKurin, a leader in innovative blood culture collection technology, is celebrating a longstanding partnership with Crouse Hospital. This collaboration marks a new era in clinical advancements, aiming to significantly reduce blood culture contamination (BCC) rates and improve patient care.
Beginning in 2012, Crouse Hospital’s initiative to combat contamination saw department-wide education, restricted blood culture draws, and rigorous compliance monitoring. These efforts yielded a modest reduction in contamination rates.
In January 2017, seeking further improvement, the emergency department re-evaluated its blood culture protocol and adopted the Kurin blood culture collection set. Michael Allain, MS, RN, ACNS-BC, CCRN, Crouse Hospital Clinical Nurse Specialist Emergency Services, noted, “We have trialed waste tubes, education, and multiple diversion devices on the market to aid in the further reduction of BCCs. Kurin was the safest and most efficient product, causing no change in blood culture collection practice. With Kurin’s advanced technology, we observed significant reductions in contamination rates, from 1.6% to .8%. This enhances diagnostic accuracy and, after accounting for product acquisition costs, achieves cost savings of over $185,000 annually for the hospital.”
Michael and his team published their outcomes in 2018 utilizing Kurin in the Journal of Critical Nurse Specialists. Following the initial implementation in the emergency department, utilization of Kurin was expanded throughout the institution.
This partnership exemplifies how integrating cutting-edge technology with evidence-based practices can result in outstanding outcomes, even for hospitals meeting standard benchmarks. Crouse’s adoption of the Kurin device and ongoing education and compliance underscores a commitment to excellence in healthcare delivery.
Kurin’s Chief Commercial Officer, Christine Arme, expressed excitement over the collaboration, stating, “This partnership with Crouse Hospital is a testament to the power of innovation in improving patient outcomes and operational efficiency. We are proud to contribute to such meaningful advances in healthcare.”
The synergy between Kurin’s technology and Crouse Hospital’s dedication to excellence sets a new standard for managing contaminated blood cultures, potentially transforming practices nationwide.
About Kurin:
Kurin® is a privately held, certified minority-owned business dedicated to engineering better healthcare through innovative, cost-effective, clinician-approved technologies. Building on the successful invention, clinical adoption, and corporate acquisition of CUROS™ disinfecting port protectors, Kurin is focusing its depth of experience in medical device engineering, marketing, sales, and service to tackle the persistent and costly fallout of false positive blood cultures. With agile development capabilities and intense customer focus, the Kurin Blood Collection Systems have quickly gained caregiver acceptance, have been proven to sideline skin contaminants during blood culture collection, and have become the market-leading solution for blood culture collection best practice.
About Crouse Hospital:
Today’s Crouse Health reflects a rich history of innovation, visionary leadership and strong community support. As the only independent, locally governed hospital in Syracuse, Crouse has been serving the community and region since 1887. Crouse is licensed for 506 acute-care adult beds and 57 bassinets, and serves more than 23,000 inpatients, 62,000 emergency services visits and more than 600,000 outpatients a year from a 16-county area in Central and Northern New York.
Crouse is a top 10 area employer, with 3,500 employees and a medical staff of more than 900 physicians. Crouse is Central New York’s largest provider of maternity care services, delivering 3,800 babies annually, and is the designated regional referral center for high-risk neonatal intensive care (NICU) services for 1,000 premature and critically ill newborns from across the region annually.
This partnership heralds a brighter future for healthcare providers and patients, driven by shared values of innovation and precision.
Reducing the Blood Culture Contamination Rate in the Emergency Department: A Good Start
/in Clinical StudiesE Browne, M Russell, G Kalukondanahally, M Abbas, O Keane, and H McDermott
Beaumont Hospital, Dublin, Ireland
Poster number: P1939
Background
Blood culture is the primary diagnostic test available to detect bloodstream infection1, however contamination during collection remains a significant issue
Objectives
Aim of the quality improvement project: reduce blood culture contamination rate in Beaumont Hospital ED by implementation of Kurin Lock® device for blood culture collection
Methods
Kurin Lock® introduced on pilot basis (8th May to 2nd July 2024)
↓
130 ED healthcare workers (HCWs) trained in device use
↓
HCWs document blood culture collection
↓
Weekly form collection and data entry
↓
Incentive
↓
Blood culture contamination data sourced from Beaumont surveillance database
↓
Weekly comparison of contamination rates
Results
A total of 768 blood cultures were collected in ED over 8 weeks (see figure 2)
Discussion
Implementation of the Kurin® Lock in Beaumont Hospital ED resulted in a significant reduction in blood culture contamination rates compared to the traditional collection method.
Read full text here
REFERENCES:
1 “Investigation of blood cultures (for organisms other than Mycobacterium species)” (UK Standards for Microbiology Investigations, PHE 2019)
2 Doern, GV et al “Practical Guidance for Clinical Microbiology Laboratories: A Comprehensive Update on the Problem of Blood Culture Contamination and a Discussion of Methods for Addressing the Problem”. Clinical Microbiology Reviews 2020; Oct 30;33(1).
3 Palavecino, EL et al “Laboratory approaches to determining blood culture contamination rates: an ASM Laboratory Practices Subcommittee report”. Journal of Clinical Microbiology 2024; Feb 14;62(2)
4 CLSI. Principles and Procedures for Blood Cultures. 2nd Ed. CLSI Guideline M47. Clinical and Laboratory Standards Institute; 2022.
5 “Kurin Lock for blood culture collection” (NICE guidance, April 2024)
6 Alahmadi, YM et al “Clinical and economic impact of contaminated blood cultures within the hospital setting”. Journal of Hospital Infection 2011; Mar 77(3):233-6